Tag: therapeutic equivalence
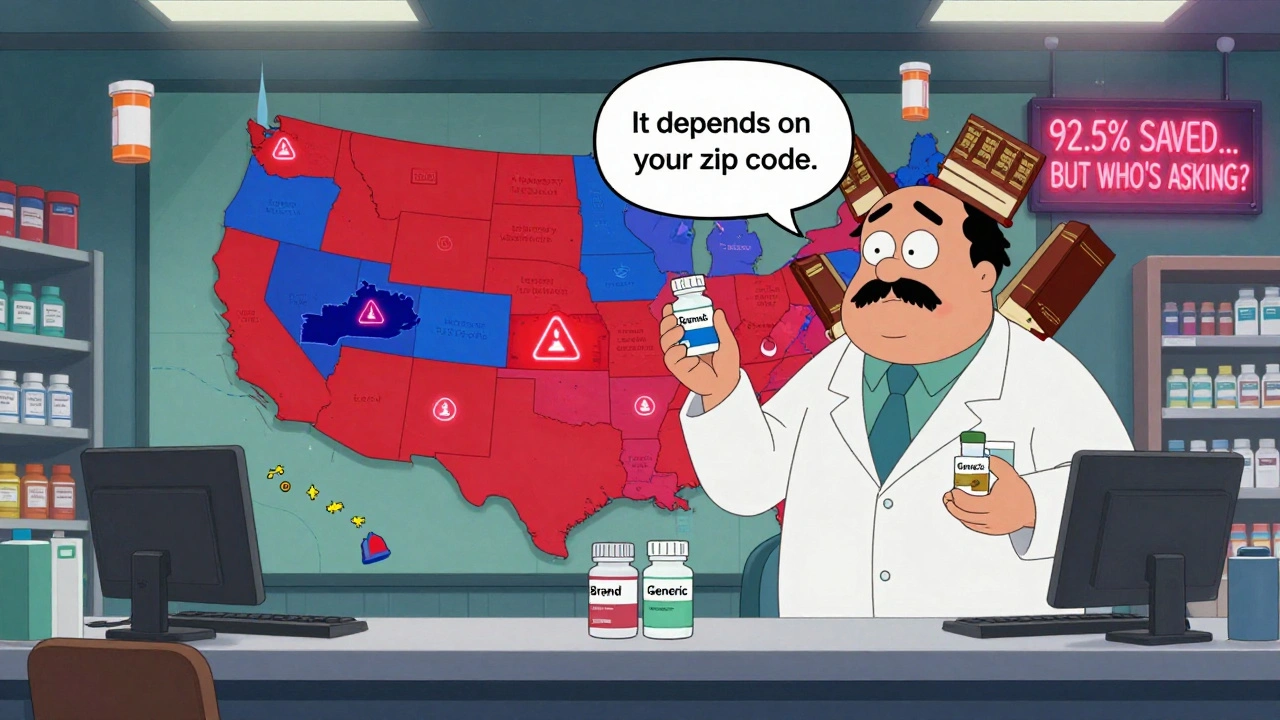
State Laws on Generic Drug Substitution: How Rules Vary Across the U.S.
State laws on generic drug substitution vary widely across the U.S., affecting how pharmacists replace brand-name drugs with cheaper generics. These rules impact cost, safety, and patient trust-especially for critical medications.