When your prescription switches from a brand-name drug to a generic version, you might notice a change in the pill’s color, shape, or packaging. If you’re worried, you’re not alone. But what if the generic you’re getting isn’t just any generic-it’s an authorized generic? That’s not a cheaper knockoff. It’s the exact same medicine, made in the same factory, with the same ingredients, just sold without the brand name on the label. And it can save you $15 to $30 per prescription.
Authorized generics aren’t new, but they’re still misunderstood. Many patients think all generics are the same. They’re not. An authorized generic is the brand-name drug itself, produced by the original manufacturer and sold under a different label. It’s approved under the same FDA application as the brand, so there’s no guesswork about safety or effectiveness. If your doctor prescribed Lipitor, and you get a pill that looks different but says "atorvastatin," it might still be the exact same tablet-just without the Pfizer logo.
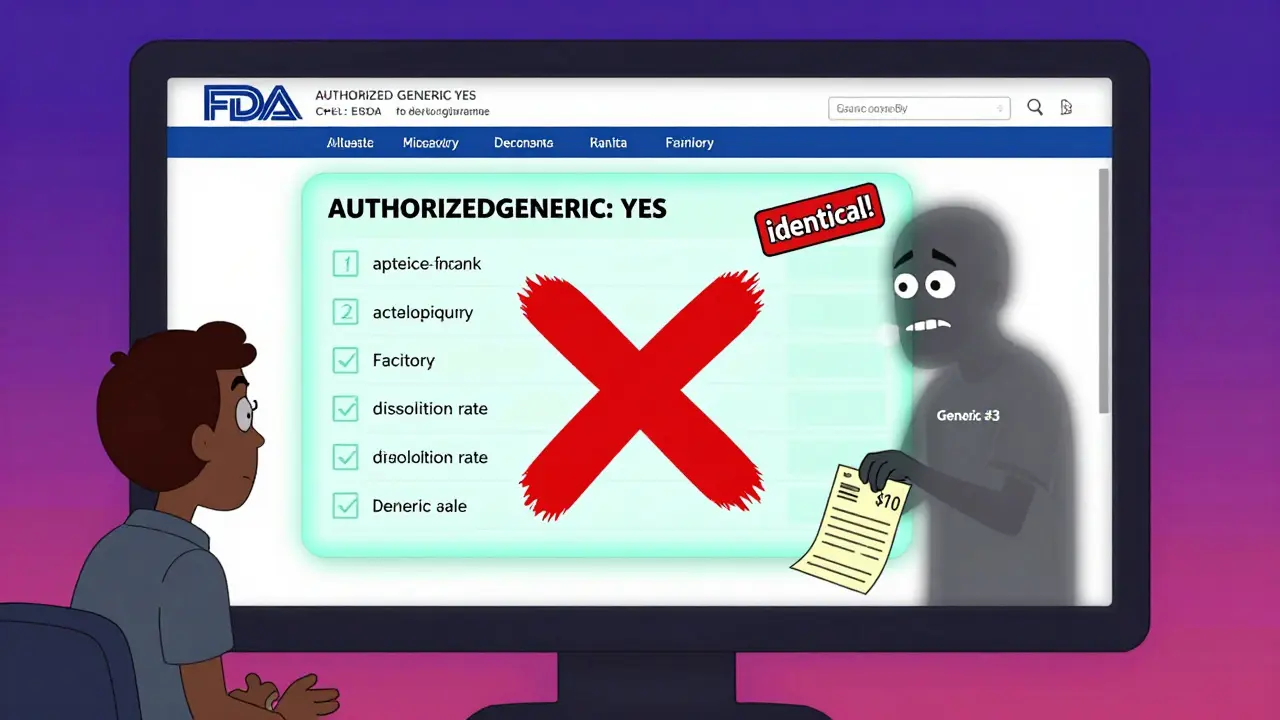
How Authorized Generics Are Different
Traditional generics must prove they work the same as the brand through bioequivalence studies. That’s a rigorous process, but it doesn’t mean they’re identical. They might use different fillers, dyes, or coatings. For most people, that’s fine. But if you’re sensitive to certain dyes, or you’re on multiple medications with complex interactions, even small differences can matter.
Authorized generics skip that step. They’re made under the brand’s original New Drug Application (NDA). That means:
- Same active ingredient
- Same inactive ingredients
- Same manufacturing facility
- Same quality control
- Same pill shape, size, and dissolution rate
The only difference? The packaging. No brand name. No fancy logo. Just the generic name on the bottle. That’s why some authorized generics look almost identical to the brand version-because they are.
The FDA keeps a public list of authorized generics updated every quarter. You can find it on their website under the Orange Book. If you’re unsure whether the generic you’re getting is authorized, check there. Don’t assume every generic is the same.
Why Authorized Generics Are Cheaper
Brand-name companies don’t give away profits. But they do use authorized generics as a tool to stay competitive. When a brand’s patent expires, the first generic company to file gets 180 days of exclusive rights to sell the generic. That’s meant to encourage competition. But here’s the twist: the brand company can launch its own authorized generic during that 180-day window-and it often does.
Why? Because if they don’t, they lose market share. By introducing their own authorized version, they keep customers who are loyal to the brand but want to save money. And because they’re the original manufacturer, they can price it lower than the first generic. Studies show that when an authorized generic enters the market:
- Retail prices drop by 4% to 8%
- Wholesale prices drop by 7% to 14%
- Patients pay 8.4% to 18.2% less on average
In 2022, Medicare Part D data showed that 78% of beneficiaries were automatically switched to authorized generics when available. That’s because insurance plans prefer them-they’re cheaper, and they’re just as effective.
What to Do When You’re Switched
Let’s say you’ve been taking a brand-name drug for years. One day, your pharmacy calls and says, "We’re giving you a different pill." You might panic. Don’t. Here’s what to do:
- Check the label-Look for the generic name (e.g., "metformin") and the manufacturer. If it’s made by the same company that makes the brand (like Pfizer, AstraZeneca, or AbbVie), it’s likely an authorized generic.
- Compare the pill-Does it look almost identical? Same color, same markings? That’s a strong sign. Authorized generics often keep the same appearance because they’re the same tablet.
- Ask your pharmacist-They can tell you if it’s an authorized generic. You can also ask: "Is this made by the same company that makes the brand?"
- Verify with the FDA-Go to the FDA’s authorized generic list. Search by the brand name. If it’s listed, you’re getting the real thing.
Some patients report confusion when the pill looks different. That’s usually because the authorized generic uses a different coating or shape-still the same formula, just not identical in appearance. Pharmacists say about 65% of patient questions are about looks, not safety. Reassure yourself: if it’s authorized, it’s not a different drug. It’s the same one, just unlabeled.

Insurance and Cost Savings
Your insurance plan doesn’t care if it’s branded or authorized. It just wants to pay less. So if an authorized generic exists, your plan will almost always require you to use it. That’s not a trick-it’s a cost-saving move.
Most plans have a tiered system. Brand-name drugs are often on Tier 3 or 4. Authorized generics are usually on Tier 1 or 2. That means:
- Lower copay
- No prior authorization needed
- No step therapy
For example, if your brand-name drug costs $60 per month with a $40 copay, the authorized generic might cost $25 with a $10 copay. That’s $30 saved per month. Over a year? $360.
Some patients worry their insurance will deny coverage if they ask for the brand. That’s rare. If you’re on a specialty drug, your doctor might need to explain why the brand is necessary-but if an authorized generic exists, insurers rarely approve the brand unless there’s a clear medical reason.
What to Watch Out For
Not every generic you get is an authorized one. Some are made by third-party companies. Those are still safe, but they might have different inactive ingredients. If you have allergies to certain dyes (like FD&C Red No. 40) or fillers (like lactose), check the inactive ingredients list on the package.
Another thing to watch: timing. Authorized generics often appear right when the brand’s patent expires-or even during the 180-day exclusivity window for the first generic. If you’re switching right after the brand goes generic, you’re likely getting the authorized version.
Some people worry that authorized generics discourage competition. That’s a valid concern. The FTC found that when brand companies launch their own generics, it can delay other generic manufacturers from entering the market. But for patients? The immediate benefit is lower prices and consistent quality.
Adherence and Safety
One of the biggest myths is that switching to a generic causes people to stop taking their meds. Studies show the opposite. When patients switch to authorized generics, adherence rates stay around 85%-the same as when they were on the brand.
Why? Because they’re the same medicine. No change in how it works. No change in side effects. No change in dosage. You take it the same way. You feel the same way. You just pay less.
For patients with chronic conditions-like high blood pressure, diabetes, or depression-staying on medication matters. Authorized generics help with that. They remove the cost barrier without introducing uncertainty.
How to Stay Informed
The FDA updates its authorized generic list every three months. You can find it at www.fda.gov/drugs/drug-approvals-and-databases/orange-book. Search by brand name. If it’s listed, the authorized version is available.
Ask your pharmacist to flag your prescriptions for authorized generics. Many pharmacy systems now automatically alert providers when an authorized version is dispensed.
Keep a list of your medications and their manufacturers. If you notice your pill looks different, check the name on the bottle. If it’s made by the same company as the brand, you’re still getting the same product.
Don’t be afraid to ask: "Is this an authorized generic?" It’s a smart question. And the answer might save you money-without sacrificing safety or effectiveness.







jared baker
March 16, 2026 AT 14:43Pro tip: Ask your pharmacist to flag your script for authorized generics. Most pharmacies can do it automatically now.
Michelle Jackson
March 18, 2026 AT 02:38Suchi G.
March 19, 2026 AT 02:31becca roberts
March 19, 2026 AT 08:22But seriously - this is genius. Insurance companies love it. Patients love it. The only ones who lose? The ones who still think brand = better. Spoiler: they’re not paying for quality. They’re paying for a logo.
Paul Ratliff
March 19, 2026 AT 14:19Gaurav Kumar
March 19, 2026 AT 22:35David Robinson
March 21, 2026 AT 03:49Also, stop pretending patients are dumb. We notice when the pill looks different. We Google it. We ask questions. We’re not helpless.
Jeremy Van Veelen
March 22, 2026 AT 10:34Also - if you’re not checking the manufacturer, you’re leaving money on the table. And dignity. And peace of mind.