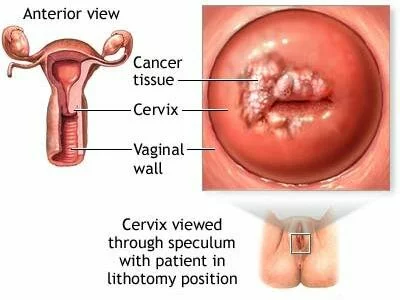
The FDA today approved Cervarix, a new vaccine to prevent cervical cancer and precancerous lesions caused by human papillomavirus (HPV) types 16 and 18. The vaccine is approved for use in girls and women ages 10 years through 25 years. Read the rest of this entry »
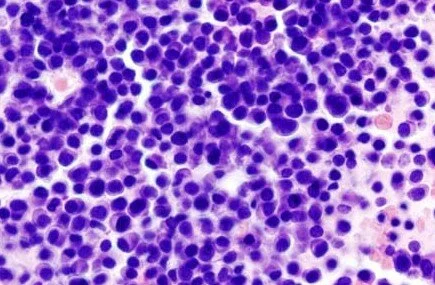
The initial data from a Phase III study using the Revlimid in multiple myeloma patients who had undergone stem cell transplant reported that the trial had met its primary endpoint of a statistically significant improvement in time to disease progression. Read the rest of this entry »
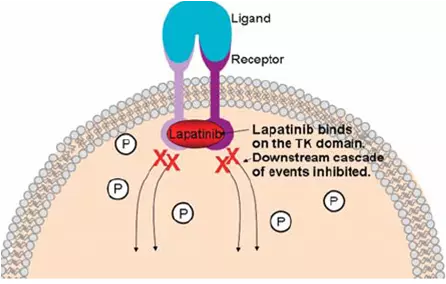
UK’s drug watchdog National Institute for Clinical Excellence (NICE) has advised against public funding of Tyverb® (lapatinib) on grounds of cost. The drug is a treatment for an aggressive form of advanced breast cancer (ErbB2-positive). Drug makers GSK say they are considering an appeal against the decision. Read the rest of this entry »




Most Commented